Into the Chloroplast: How Photosynthesis Works
Article objectives
Life requires photosynthesis for fuel and for the oxygen to burn that fuel. Since the Industrial Revolution (late 18\(^{th}\) and early 19\(^{th}\) centuries), we humans have relied on products of ancient photosynthesis for enormous quantities of fossil fuel energy. And, knowingly or not, we have also benefited from photosynthesis to remove the carbon dioxide produced when we burn those fuels. So it may not surprise you that biologists have studied this critical process in great detail.
A single chemical reaction represents the overall process of photosynthesis as demonstrated in the equation below.

Although photosynthesis may seem straightforward in this form, such simplicity is deceiving for two reasons. First, the equation above summarizes dozens of individual chemical reactions involving many intermediate compounds. And second, just discovering major players like \(CO_{2}\) and \(O_{2}\) was challenging, because our ordinary senses cannot detect these molecules in “thin air!”
How do we know that the chemical reaction in photosynthesis really happens? Two famous historical experiments help us begin to understand this process.

Figure 1: In the \(17^{th}\) century, Jan Van Helmont, a Flemish chemist, physiologist, and physician, weighed and potted a willow tree, showing that plants do not get food from the soil.
In the \(17^{th}\) century, people who thought about it at all assumed that plants get their food from the soil. Many people, encouraged by sellers of “plant food,” still do. In 1638, Jan Baptist Van Helmont planted a 5 pound willow tree, like the one shown in Figure 1, in a 200 pound tub of soil. After 5 years of watering the plant, he weighed both again. The willow had gained over 160 pounds, but the soil had lost only 2 ounces. Van Helmont concluded that plants do not get their materials from soil, and inferred that they grow using materials from water (which he did not measure). As you know now, he was half right. Although soil provides important nutrients to plants, it supplies neither the energy nor the vast majority of the materials to build the plant. We must excuse him, because no one in the 17\(^{th}\) century knew that carbon atoms form the basis of life, or that they float around in air in the form of carbon dioxide.
In the late 1770s, minister and natural philosopher Joseph Priestley burned a candle in a jar of air and observed that the candle burned out long before it ran out of wax. A similar experiment with a mouse resulted in the mouse’s death. Priestley suggested that animals, like candles, “injure” the air. Adding a mint plant, as shown in Figure 2, however, “restored” the air which had been “injured” by the mouse or the candle. Only later, after many chemistry experiments, did Priestley publish his discovery of “dephlogisticated air.” But in his studies of mice, plants, and candles, he had shown that plants produce, and animals consume, oxygen gas.

During the 20\(^{th}\) century, we learned that photosynthesis involves much more than just the three reactants, the three necessary conditions, and the two products shown in the equation. Using powerful microscopes, we’ve narrowed the process to one type of organelle within the plant – the chloroplast. In the next section, you will learn in more detail just how plants, algae, and photosynthetic bacteria make food for us all “from thin air.” First, let’s look at the organelle in which the drama of photosynthesis takes place and meet some of the key actors.
Chloroplasts: Theaters for Photosynthesis
If you examine a single leaf of the aquatic plant Elodea, shown in Figure 3, under a microscope, you will see within each cell dozens of small green ovals. These are chloroplasts, the organelles which conduct photosynthesis in plants and algae. Chloroplasts closely resemble some types of bacteria and even contain their own circular DNA and ribosomes. In fact, the endosymbiotic theory holds that chloroplasts were once independently living bacteria (prokaryotes). So when we say that photosynthesis occurs within chloroplasts, we speak not only of the organelles within plants and algae, but also of some bacteria – in other words, virtually all photosynthetic autotrophs.

Both chloroplasts and photosynthetic bacteria contain neat stacks (grana) of flattened sacshaped membrane compartments (thylakoids), made in turn of elaborate and highly organized patterns of molecules which conduct photosynthesis, as shown in Figure 4. In addition to enzymes, two basic types of molecules - pigments and electron carriers – are key players.

Pigment molecules, often arranged together with proteins in large, complex photosystems, absorb specific wavelengths of light energy and reflect others; therefore, they appear colored. The most common photosynthetic pigment is chlorophyll, which absorbs blue-violet and red wavelengths of light, and reflects green (Figure 5 and Figure 6). Accessory pigments absorb other colors of light and then transfer the energy to chlorophyll. These include xanthophylls (yellow) and carotenoids (orange).


Electron carrier molecules are usually arranged in electron transport chains (ETCs). These accept and pass along energy-carrying electrons in small steps (Figure 7). In this way, they produce ATP and NADPH, which temporarily store chemical energy. Electrons in transport chains behave much like a ball bouncing down a set of stairs – a little energy is lost with each bounce. However, the energy “lost” at each step in an electron transport chain accomplishes a little bit of work, which eventually results in the synthesis of ATP.
Now that you’ve met some of the key players and explored the theater, let’s put them together to see how the process unfolds. We will divide the process into two basic sets of reactions, known as the light reactions and the Calvin cycle, which uses carbon dioxide. As you study the details, refer frequently to the chemical equation of photosynthesis. In the first stage, you’ll discover how chloroplasts transform light energy, and why we owe our ability to breathe to plants!

Photosynthesis Stage I: The Light Reactions: in which Chloroplasts Capture Sunlight Chemical Energy…
Every second, the sun fuses over 600 million tons of hydrogen into 596 tons of helium, converting over 4 tons of helium (4.3 billion kg) into light and heat energy. Countless tiny packets of that light energy travel 93 million miles (150 million km) through space, and about 1% of the light which reaches the Earth’s surface participates in photosynthesis. Light is the source of energy for photosynthesis, and the first set of reactions which begin the process requires light – thus the name, Light Reactions, or Light-dependent Reactions.
When light strikes chlorophyll (or an accessory pigment) within the chloroplast, it energizes electrons within that molecule. These electrons jump up to higher energy levels; they have absorbed or captured, and now carry, that energy. High-energy electrons are “excited.” Who wouldn’t be excited to hold the energy for life?
…And Change the Rules of Chemistry for Life!
The excited electrons leave chlorophyll to participate in further reactions, leaving the chlorophyll “at a loss”; eventually they must be replaced. That replacement process also requires light, working with an enzyme complex to split water molecules. In this process of photolysis (“splitting by light”), \(H_{2} O\) molecules are broken into hydrogen ions, electrons, and oxygen atoms. The electrons replace those originally lost from chlorophyll. Hydrogen ions and the high-energy electrons from chlorophyll will carry on the energy transformation drama after the Light Reactions are over.
The oxygen atoms, however, form oxygen gas, which is a waste product of photosynthesis (Figure 8). The oxygen given off supplies most of the oxygen in our atmosphere. Before photosynthesis evolved, Earth’s atmosphere lacked oxygen altogether, and this highly reactive gas was toxic to the many organisms living at the time. Something had to change! Most contemporary organisms rely on oxygen for efficient respiration. So plants don’t just “restore” the air, as Priestley suggested. They also had a major role in creating it!
To summarize, chloroplasts “capture” sunlight energy in two ways. Light “excites” electrons in pigment molecules, and light provides the energy to split water molecules, providing more electrons as well as hydrogen ions.

Now let’s follow those excited electrons…
How Do Chloroplasts Convert Light Energy to Chemical Energy?
Excited electrons which have absorbed light energy are unstable. However, the highly organized electron carrier molecules embedded in chloroplast membranes order the flow of these electrons, directing them through electron transport chains (ETCs). At each transfer, small amounts of energy released by the electrons are captured and put to work or stored. Some is also lost as heat with each transfer, but overall the light reactions are extremely efficient at capturing light energy and transforming it to chemical energy.
Two sequential transport chains harvest the energy of excited electrons, as shown in Figure 9.
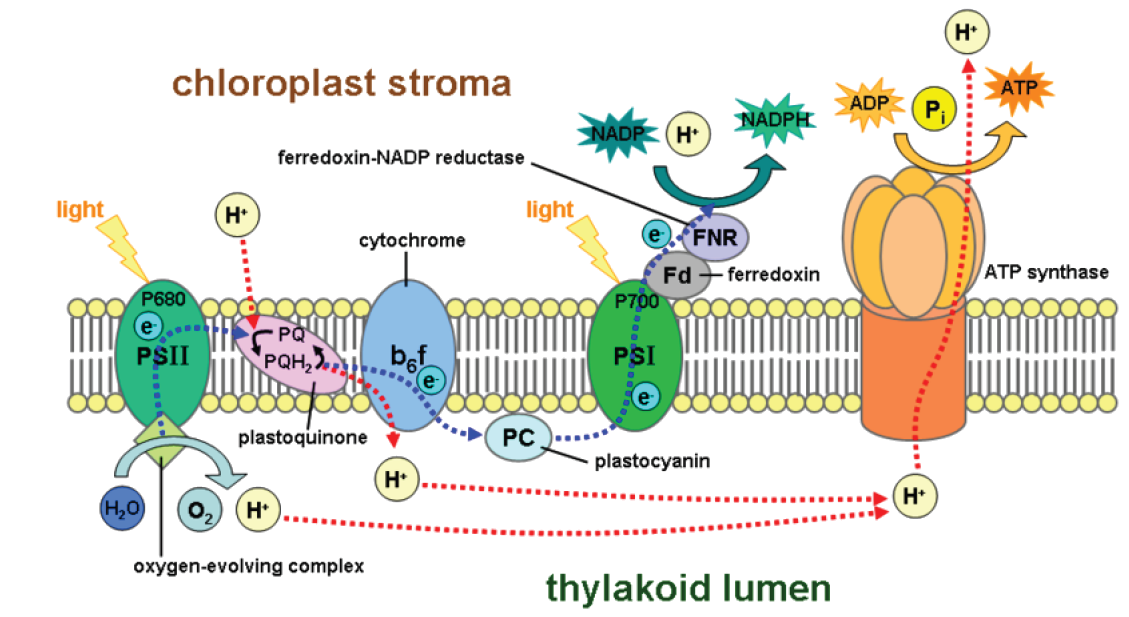
\({1}\) First, they pass down an ETC which captures their energy and uses it to pump hydrogen ions by active transport into the thylakoids. These concentrated ions store potential energy by forming a chemiosmotic or electrochemical gradient – a higher concentration of both positive charge and hydrogen inside the thylakoid than outside. (The gradient formed by the H\(^{+}\) ions is known as a chemiosmotic gradient.) Picture this energy buildup of H\(^{+}\) as a dam holding back a waterfall. Like water flowing through a hole in the dam, hydrogen ions “slide down” their concentration gradient through a membrane protein which acts as both ion channel and enzyme. As they flow, the ion channel/enzyme ATP synthase uses their energy to chemically bond a phosphate group to ADP, making ATP.
\({2}\) Light re-energizes the electrons, and they travel down a second electron transport chain (ETC), eventually bonding hydrogen ions to NADP\(^{+}\) to form a more stable energy storage molecule, NADPH. NADPH is sometimes called “hot hydrogen,” and its energy and hydrogen atoms will be used to help build sugar in the second stage of photosynthesis.
NADPH and ATP molecules now store the energy from excited electrons – energy which was originally sunlight – in chemical bonds. Thus chloroplasts, with their orderly arrangement of pigments, enzymes, and electron transport chains, transform light energy into chemical energy. The first stage of photosynthesis – light-dependent reactions or simply “light reactions” – is complete.
Photosynthesis Stage II: The Calvin Cycle - Making Food “From Thin Air”
You’ve learned that the first, light-dependent stage of photosynthesis uses two of the three reactants - water and light - and produces one of the products - oxygen gas (a waste product of this process). All three necessary conditions are required – chlorophyll pigments, the chloroplast “theater,” and enzyme catalysts. The first stage transforms light energy into chemical energy, stored to this point in molecules of ATP and NADPH. Look again at the overall equation below. What is left?

Waiting in the wings is one more reactant – carbon dioxide, and yet to come is the star product which is food for all life – glucose. These key players perform in the second act of the photosynthesis drama, in which food is “made from thin air!”
The second stage of photosynthesis can proceed without light, so its steps are sometimes called “light-independent” or “dark” reactions. Many biologists honor the scientist, Melvin Calvin, who won a 1961 Nobel Prize for working out this complex set of chemical reactions, naming it the Calvin Cycle.
The Calvin Cycle has two parts. First carbon dioxide is ”fixed.” Then ATP and NADPH from the Light Reactions provide energy to combine the fixed carbons to make sugar.
Carbon Dioxide is “Fixed”
Why does carbon dioxide need to be fixed? Was it ever broken?
Life on Earth is carbon-based. Organisms need not only energy but also carbon atoms for building bodies. For nearly all life, the ultimate source of carbon is carbon dioxide (\(CO_{2}\)), an inorganic molecule. \(CO_{2}\), as you saw in Figure 11, makes up .038% of the Earth’s atmosphere.
Animals and most other heterotrophs cannot take in \(CO_{2}\) directly. They must eat other organisms or absorb organic molecules to get carbon. Only autotrophs can build low energy inorganic \(CO_{2}\) into high-energy organic molecules like glucose. This process is carbon fixation.
Plants have evolved three pathways for carbon fixation. The most common pathway combines one molecule of \(CO_{2}\) with a 5-carbon sugar called ribulose biphosphate (RuBP). The enzyme which catalyzes this reaction (nicknamed RuBisCo) is the most abundant enzyme on earth! The resulting 6-carbon molecule is unstable, so it immediately splits into two 3-carbon molecules. The 3 carbons in the first stable molecule of this pathway give this largest group of plants the name “C-3.”
Dry air, hot temperatures, and bright sunlight slow the C-3 pathway for carbon fixation. This is because stomata, tiny openings under the leaf which normally allow \(CO_{2}\) to enter and \(O_{2}\) to leave, must close to prevent loss of water vapor (Figure 11). Closed stomata lead to a shortage of \(CO_{2}\). Two alternative pathways for carbon fixation demonstrate biochemical adaptations to differing environments.

Plants such as corn solve the problem by using a separate compartment to fix \(CO_{2}\). Here \(CO_{2}\) combines with a 3-carbon molecule, resulting in a 4-carbon molecule. Because the first stable organic molecule has four carbons, this adaptation has the name C-4. Shuttled away from the initial fixation site, the 4-carbon molecule is actually broken back down into \(CO_{2}\), and when enough accumulates, Rubisco fixes it a second time! Compartmentalization allows efficient use of low concentrations of carbon dioxide in these specialized plants.
Cacti and succulents such as the jade plant avoid water loss by fixing \(CO_{2}\) only at night. These plants close their stomata during the day and open them only in the cooler and more humid nighttime hours. Leaf structure differs slightly from that of C-4 plants, but the fixation pathways are similar. The family of plants in which this pathway was discovered gives the pathway its name, Crassulacean Acid Metabolism, or CAM (Figure 12). All three carbon fixation pathways lead to the Calvin Cycle to build sugar.

How Does the Calvin Cycle Store Energy in Sugar?
As Melvin Calvin discovered, carbon fixation is the first step of a cycle. Like an electron transport chain, the Calvin cycle, shown in Figure 13, transfers energy in small, controlled steps. Each step pushes molecules uphill in terms of energy content. Recall that in the electron transfer chain, excited electrons lose energy to NADPH and ATP. In the Calvin Cycle, NADPH and ATP formed in the light reactions lose their stored chemical energy to build glucose.
Use the diagram below to identify the major aspects of the process: • the general cycle pattern • the major reactants • the products
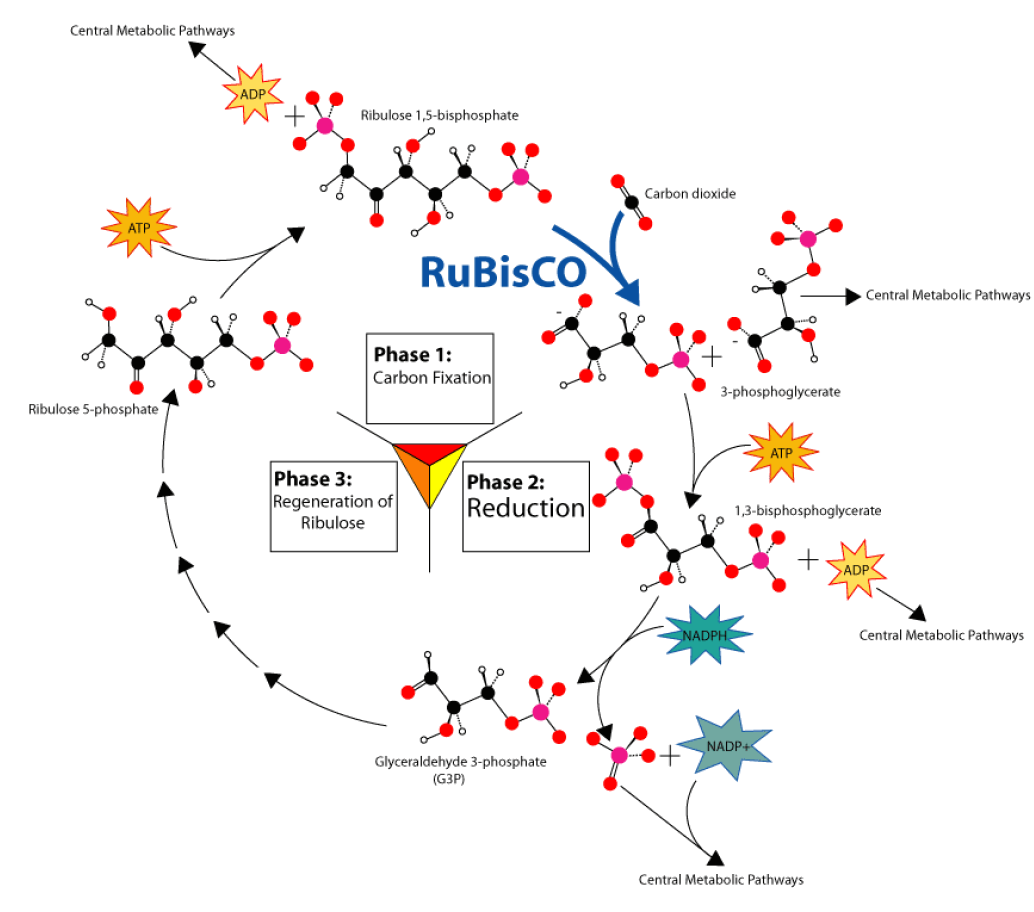
First, notice where carbon is fixed by the enzyme nicknamed Rubisco. In C-3, C-4, and CAM plants, \(CO_{2}\) enters the cycle by joining with 5-carbon ribulose bisphosphate to form a 6-carbon intermediate, which splits (so quickly that it isn’t even shown!) into two 3-carbon molecules.
Now look for the points at which ATP and NADPH (made in the light reactions) add chemical energy (“Reduction” in the diagram) to the 3-carbon molecules. The resulting “half-sugars” can enter several different metabolic pathways. One recreates the original 5- carbon precursor, completing the cycle. A second combines two of the 3-carbon molecules to form glucose, universal fuel for life.
The cycle begins and ends with the same molecule, but the process combines carbon and energy to build carbohydrates – food for life.
So – how does photosynthesis store energy in sugar? Six “turns” of the Calvin cycle use chemical energy from ATP to combine six carbon atoms from six \(CO_{2}\) molecules with 12 “hot hydrogens” from NADPH. The result is one molecule of glucose, \(C_{6} H_{12} O_{6}\).
Images courtesy of:
CK-12 Foundation. CC-BY-SA. http://en.wikipedia.org/wiki/Image:Par_action_spectrum.gif. CC-BY-SA 2.0.
CK-12 Foundation. Joseph Priestly’s bell jar experiment. CC-BY-SA.
http://www.flickr.com/photos/dropandroll/368377017. Creative Commons.
http://commons.wikimedia.org/wiki/Image:Chloroplast-new.jpg. Public Domain.
http://en.wikipedia.org/wiki/Image:Chlorophyll_structure.png. Public Domain.
http://commons.wikimedia.org/wiki/File: Chromatography_of_chlorophyll_-_Step_6.jpg. CC-BY-SA 2.5.
http://en.wikipedia.org/wiki/Image:The_Earth_seen_from_Apollo_17.jpg. Public Domain.
http://en.wikipedia.org/wiki/Image:Thylakoid_membrane.png. Public Domain.
Alex Costa,Gross L, PLoS Biology Vol. 4/10/2006, ed. 358. http://dx.doi.org/10.1371/journal.pbio.0040358. Public Domain,CC-BY.
http://www.flickr.com/photos/lobo235/76154752/ http://www.flickr.com/photos/jylcat/562393266/. CC-BY 2.0.
Mike Jones. Overview of the Calvin Cycle Pathway.. CC-BY-SA 2.5.